Real-time Chemical Characterization of Atmospheric Particulate Matter in China a Review
1. Introduction
Atmospheric pollutants are a circuitous system composed of various gaseous pollutants and particulate pollutants, which play an of import role in the radiations balance of the atmospheric arrangement, and likewise touch on the efficient utilize of energy, i.e., solar energy [ane,ii,three]. At the aforementioned time, atmospheric particulate thing pollution has a very important impact on human being health, atmospheric climate and radiation. It is an important air pollutant affecting human health and reducing visibility [4,5]. Studies have already shown that PM2.v tin easily absorb other pollutants, including heavy metal, organic chemicals, bacteria and viruses. Due to the small particle size of PM2.5, information technology can penetrate deep into the lungs, and long-term exposure to polluted environments volition increment the incidence of cardiovascular and cardiopulmonary diseases [6,7,8,9]. The chemical limerick of atmospheric particulate matter is circuitous and varies greatly in dissimilar regions, but it is basically composed of carbonaceous substances (Elemental Carbon and Organic Carbon), water-soluble ions and diverse inorganic heavy metal elements [ten,11]. Heavy metal elements in atmospheric particles will seriously affect the growth of plants [12,13]. Particulate matter will also carry heavy metal elements into the human alveoli, which is more harmful to humans. Organic carbon and elemental carbon in aerosols are the main causes of visible low-cal digestion, which reduces atmospheric visibility and causes extreme weather [14]. At the same fourth dimension, aerosol particles are also the principal components of cloud condensation nuclei, which can touch on the lifetime and optical properties of the cloud, and indirectly affect the climate [15].
Some scholars' researches in the field of atmospheric aerosols have made peachy contributions to the governance of the atmospheric environment. Cheng [16] et al. nerveless PM2.five, tested their inorganic elements, and used PMF models to written report the sources of PM2.v in Wuhan city, and institute that motor vehicle emissions and secondary ion compound pollution touch PM2.v's contribution is more than 50%. Querol [17] et al. used Chief Component analysis to analyze the sources of PM10 in the atmospheric environment of Wuhan. The results showed that industry behaviors such as cement and steel product, were the largest pollution sources of PM10 in industrial areas, accounting for 34%, followed by coal burning (20%) and the contribution of the movement of regional pollution sources (sixteen%). In urban areas, coal has the largest contribution rate (31%), followed by the transfer of regional pollution sources (28%), with traffic sources accounting for 16%. Huang [18] et al. analyzed the causes and sources of air pollution in Wuhan in summertime and winter, the results bear witness that the summer air pollution process in Wuhan was profoundly affected past the use of solvent coatings, motor vehicle exhaust emissions and industrial emissions of VOCs. Environmental pollution in winter is mainly affected by secondary aerosol sources, coal-burning industrial sources and motor vehicle sources. At the same time, regional manual as well has a certain bear upon on the occurrence of polluted weather in Wuhan: the polluted air masses in summertime and wintertime come from the southeast and northwest of Hubei Province, respectively. Chen [19] et al. studied the chemical composition of PM2.5 in Harbin and analyzed the elements in PM2.5 by enrichment factor analysis. It was found that Cr, Ni, Cu, Br and Zn were highly enriched, indicating that these elements were seriously affected by man activities. Zhang [20] et al. studied the potential sources and source regions of PM1 and PM2 in Beijing using PMF model, Backward Trajectory model and Potential Source Contribution part model. The results evidence that secondary droplets and coal combustion, vehicle industry and biomass combustion are the main sources of atmospheric particulate matter.
The higher up research results are based on the typical areas of the city, and the selections of research sites belong to the areas with high population density, only the popularization of these enquiry conclusions is limited, especially for some other typical areas with different pollution conditions. In this study, the atmospheric environment nether various meteorological conditions in the Huangjiahu surface area of Wuhan is sampled, and its chemic composition and source characteristics are studied. The research results have certain guiding significance for many industries such as environmental monitoring, energy utilization, wellness care, transportation and so on.
two. Experimental Methods
two.1. Sampling Location
Wuhan has a long history of industrial development. With the continuous evolution of the economy, its urban construction scale is becoming larger and larger. In recent years, the research on aerosol particles in the urban area of Wuhan has gradually go one of the research hotspots in this field. The location of this written report is near the Baishazhou Avenue of Wuhan city, which is ane of the busiest transportation roads in Wuhan. Although the relevant departments have made slap-up progress in environmental handling here in recent years (adding dark-green isolation belt and other measures), there is all the same droplets particle pollution, especially in winter and spring, with the increment of pollutant content and the interaction betwixt various pollutant emission sources (automobile frazzle, dust and industrial emission, etc.). Although many scholars and research teams have studied the source analysis of droplets particles in Wuhan, due to regional differences, as well every bit differences in the traffic environment and the natural environment, it is more hard to clarify the pollutant source. Therefore, it is necessary to study the source analysis of particulate matter in the typical area of the Huangjiahu area.
Effigy 1 shows the specific location of the sampling indicate. It is located on the environmental test outdoor platform (114.24° East, 30.44° N) on the roof of the third floor of the 5th didactics edifice of Wuhan University of science and technology. Horizontal height is 15 m away from the ground and is less disturbed past ground dust. At that place is a wide green plant coverage area between Baishazhou Avenue, Huangjiahu West Route and the sampling indicate. The selection of this betoken is representative.
2.2. Sample Collection and Storage
The DL-6100 medium-menses atmospheric particulate matter sampler has been used for sampling. The working signal of the sampler is 100 L/min with an error of no more than than 2%. It is equipped with PM10 cutter and PM2.5 cutter, and the cutting particle size is Da50 = 2.v + 0.2 μm. The geometric standard departure of the collection efficiency is σg = 1.ii + 0.one μm. Other performance and technical indicators are in line with HJ/T93-200 regulations. The filter membrane used for sampling is the quartz fiber filter membrane, with a diameter of xc mm, and the interception efficiency of the filter membrane for 0.iii μm standard particles is non less than 99.seven%. Before sampling, the quartz filter membrane is burned in a muffle furnace at a temperature of 500 °C for 4 h. After burning, it is placed in a chamber with a constant temperature and humidity for balance for 24 h. It is besides weighed and recorded during this time. The collection time of each sample is 20 h. After the sample is collected, information technology is balanced once again in a constant temperature and humidity chamber for 24 h, is also weighed and recorded, and then placed in a closed refrigeration at 4 °C until detection.
2.3. Analysis Method
The detection instruments used in this paper include Fourier transform infrared spectrometer (FTIR), ICS-2000 Ion Chromatograph (IC), free energy dispersive 10-ray fluorescence spectrometer (XRF) to qualitatively and quantitatively clarify the functional groups, water-soluble anions and inorganic elements in the sample.
2.3.1. Fourier Transform Infrared Spectrometer Measurement
FTIR can qualitatively analyze the functional groups contained in the sample. By comparison and analyzing the infrared spectra of standard materials, it can effectively determine the types of substances contained in the sample [21,22]. In ecology analysis, FTIR tin can effectively identify substances with infrared characteristics, such as nitric oxide, nitrogen dioxide, nitrogen oxides, sulfur dioxide, chlorofluorocarbons and so on. In club to reduce the influence of filter membrane, blank filter membrane pressure sheet can be fabricated to eliminate interference. FTIR of Perkin Elmer Borderland is used to scan the sample tablet. The wavelength scanning range of the instrument is 4000–400 cm−1, the scanning times is 32, and the resolution is iv cm−1. In order to reduce the experimental mistake, each sample is tested twice. Through the analysis of FTIR, the functional groups and chemical bonds contained in the collected PM2.5 particles tin be identified, and so as to judge the types of substances in the particles. We nerveless a total of 12 sample sets from 26 April to 24 May. The sample numbers and sampling times are shown in Table 1.
2.3.2. Ion Chromatography Measurement
The water-soluble ions in PM2.5 are measured quantitatively by IC to obtain the specific content of water-soluble ions in atmospheric particles. One quarter of the filter membrane sample is used to measure out the content of h2o-soluble ions. 100 mL pure water is added to the sample to immerse the filter membrane. Comprehend it and soak it for xxx min, and then put it into an ultrasonic cleaner to vibrate and excerpt for 20 min. Subsequently the extraction process is completed, filter it with an air extraction filtration device, and and so use the filtrate with 0.45 μm aqueous microporous blue membrane syringe filter, manually inject the sample into the ion chromatograph for detection.
In this study, ICS-2000 Ion Chromatograph is used. Firstly, six different concentrations of labeled stock solutions are equipped. The standard solutions of 6 concentrations are successively injected into the ion chromatograph, and the peak surface area (or summit height) is recorded. Then, the standard curve is drawn with the superlative area of each ion as the ordinate and the standard concentration as the abscissa. Finally, the concentration of each ion is calculated according to the peak area (elevation height) of the sample. Table 2 presents the standard curves of four anions.
2.3.three. X-ray Fluorescence Spectrometry Measurement
The Ten-ray fluorescence spectrometer of Perkin Elmer Frontier energy dispersion model is used to quantitatively analyze the inorganic elements in the sample. The atoms of the elements are excited by loftier-free energy radiation to cause the transition of inner electrons, and emit X-rays with a sure special wavelength. The element content can be measured quantitatively according to this characteristic.
A circular pocketknife with a bore of 47 mm is used to cut a filter membrane disc with a diameter of 47 mm. The element content in the particulate sample is calculated according to Formula (1):
where, A represents the area loaded with particles on the filter membrane, I represents the 10-ray fluorescence intensity of the target element in the sample filter membrane, I 0 represents the X-ray fluorescence intensity of the target element in the blank filter membrane, b is the slope of the calibration curve, 5 is the sampling volume in the standard state, and ρ is the content of the target chemical element in the particle sample.
3. Research Results
three.i. Assay of General Characteristics of Pollutants in Wuhan from 2017 to 2021
The boilerplate daily concentration data of PM2.5 and PM10 in Wuhan from January 2017 to July 2021 are from Tianyun big data website [23]. The average monthly particle concentration changes during this period are shown in Effigy 2a, the average daily particle concentration changes during the sampling menstruation are shown in Figure 2b. Both PM2.five and PM10 prove obvious seasonal changes: the seasonal change of PM2.5 is mainly reflected in the higher concentration in winter and lower in spring, summer and autumn, the main tendency of seasonal variation is: Wintertime (December to February of the following year) > spring (March to May) > autumn (June to August) > summertime (September to November). The seasonal variation characteristics of PM10 are the same as PM2.5. The concentration of particulate matter in summertime is the season with lower concentration in a yr, and the seasonal variation is mainly reflected in winter > jump > autumn > summer. The main reasons for this seasonal change are every bit follows:
(1) Wuhan is a subtropical monsoon boiling climate region. In tardily jump and early summer, the cold air flow slows down and the hot air flow in the South picks upward, which will cause frequent gale and pelting or continuous cloudy and rainy weather in Wuhan. Therefore, the air quality in Wuhan is significantly improved. With the continuous rise of temperature, the surface humidity decreases, and grit is easy to exist hoisted in windy conditions, which further increases the content of atmospheric particulate matter. At the aforementioned fourth dimension, from March to May, the dusty weather in the North increased, and the particles diffused with the atmospheric circulation, which made a certain contribution to the increase of particle content in Wuhan, which may be the main reason for a short recovery of PM10 in spring;
(2) In summertime, it is hot and rainy, accompanied by potent winds, which has a good dilution upshot on the diffusion of particulate matter and reduces the residence time of particulate matter in the atmosphere. In summer, plants grow luxuriantly and the leaf area increases, which is too conducive to the adsorption of atmospheric particulate matter, resulting in the retention of atmospheric particulate affair and reducing the content of particulate matter in the air [24]. In autumn and winter, leaves wither, constitute photosynthesis decreases, and the retentivity of atmospheric particulate matter decreases accordingly. In winter, Wuhan is plainly afflicted by the winter monsoon climate, and the daytime is short, which leads to the strengthening of long wave radiation at night. Especially in the sunny and less windy climate, an atmospheric inversion layer is formed, which weakens the diffusion ability of particulate matter. In improver, the air temperature in winter is low and dry out, which is conducive to the secondary winding of ground dust. Secondly, due to the centralized heating in some areas of Wuhan in winter, the emission of pollutants has increased, resulting in a high concentration of particulate thing in winter.
iii.two. Aerosol Composition Analysis past FTIR
In Figure iii, the infrared spectra of 28 April, 30 Apr and vi May in 2021 are shown. The infrared spectra of these days include almost all the functional groups and chemical bonds contained in the samples during the sampling catamenia. The approximate limerick of the particulate matter can be obtained by analyzing infrared spectroscopy. In this study, the master groups of organic matter independent in the sample are OH and C=C. OH and C=C were found in all samples; NO3 − and And theniv 2− are the main ions in inorganic salts, which are mainly generated by some chemical reactions of NOx and And soten in the atmosphere [25]. The common oxides plant in the samples are SiOtwo, MnO2, Fe3O4 and MgO et al. And almost all samples contain SiO2. In addition to sample 6, MnO2 exists in other samples, and MgO exists in samples three, four, 5, 6, 7 and 8. NHfour + and CO3 2− were not detected during the test. The reason may be that the sample is non frozen and insulated during sample delivery, resulting in its volatilization, and other ions will not volatilize, so the specific content tin can be measured accurately.
3.3. Ion Chromatographic Analysis of Droplets Particles
According to the measurement results of Fourier transform infrared spectroscopy, So4 2−, NO3 −, Cl− and F− ions in PM2.5 were detected by IC. The concentration data of four anions are shown in Figure 4. It can be seen that the concentration of the iv ions during the sampling menstruum from large to pocket-size is: And then4 two− > NO3 − > Cl− > F−, and the mass concentrations are: 9.166 ± 4.151 μg/yard3, 2.548 ± ii.384 μg/chiliadthree, 0.658 ± 0.317 μg/m3, 0.042 ± 0.017 μg/1000iii.
SO4 2− mainly comes from the conversion of Thentwo in the temper. As the radiation intensity in summertime increases, the photochemical reaction increases, resulting in an increase in the mass concentration of Then4 two− [26]. The concentration of SO4 ii− is related to the oxidation efficiency of SO2. The Sulfur conversion rate is ordinarily expressed by SOR, and its adding formula is:
where,
is the mass concentration of SO4 ii−,
is the mass concentration of SO2. When in that location is only one pollutant in the atmosphere, SOR < 0.1; when SOR > 0.ane, it means that the And then2 in the atmosphere may undergo a photochemical reaction. Through calculation, during the sampling period, the SOR was less than 0.1, indicating that during the sampling period, the Thenii in the atmosphere may non undergo a photochemical reaction, or the intensity of the photochemical reaction may not be large.
By analyzing the correlation between the ions, we tin can get the consistency between the ion sources [27]. The correlation coefficient of So4 2− and NO3 − is 0.76, which can betoken that their source characteristics are similar, and the chief source is fossil fuel combustion. In addition, the mass ratio of SOiv 2− and NOiii − can be used to compare the contribution of stationary sources (such equally coal) and mobile sources (such equally automobile exhaust) to the amount of S and N in the atmosphere. During the sampling period, the changes in the ratio of SOfour ii− to NO3 − are shown in Figure five. The ratios are all less than one, indicating that during the sampling catamenia, the emission sources from mobile sources are less than those from stationary sources.
3.iv. Source Apportionment past XRF and Enrichment Factor Analysis
XRF was used to further clarify the elemental composition of the 12 samples collected. The analysis results are shown in the Figure 6. Amid all the detected elements, Si has the highest proportion in the sample, accounting for 53.19%, followed by Ca, Atomic number 26, S, K and other trace elements accounted for 12.85%, 9.ix%, 9.xv%, half-dozen.08% and 8.84% respectively. Figure 6a shows the proportion of each element, and Figure 6b shows the proportions of the relative contents of other trace elements. Based on the XRF assay results, the enrichment of each element was analyzed by enrichment cistron analysis.
Enrichment factor analysis is an effective means to analyze the source and caste of pollutants in the surroundings. Enrichment gene tin can reflect the enrichment degree of elements in atmospheric particulate affair relative to elements in the surface of the world's crust, and then every bit to evaluate the influence caste of natural and human being-made sources of various elements [27,28,29]. The enrichment factor method is used to analyze the elements of PM2.5 in the atmosphere to determine the enrichment pollution status of each chemical element, which can be used for more in-depth analysis. The calculation formula of the enrichment factor is equally follows:
where,
represents the enrichment gene of the element,
represents the content of the target element, and
represents the content of the reference element. By and large, depression volatile elements unremarkably exist in the earth'southward crust with less anthropogenic pollution sources, good chemical stability and high accuracy of analysis results are selected as reference elements. Fe, Al, Si, Ti and Sc are widely used internationally. In this written report, Fe element is selected as the reference element and the soil element in layer A of Hubei Province is selected as the background value of the study. If EF ≤ 1, it means no pollution, indicating that the element is mainly a natural source, such as rock weathering, dust winch, etc.; 1 < EF < 10, indicating that the element has both natural and man-made sources; EF > 10 indicates that the enrichment caste of this element is high, mainly from human being-made pollution. The higher the value of EF, the more likely it is to come from man-made pollution.
The enrichment factors are shown in Figure seven. The enrichment factors of Na, Mg, Al, Ti and V are between 0 and 1, indicating that these elements are mainly derived from materials in the earth'due south crust. The enrichment factors of Chiliad, Cr, Mn, Ni and Ba are between l and 10, and these elements are affected by both man-made sources and natural sources. The enrichment factors of Ca, Sc, Cu and Co are more often than not between 10 and 25, indicating that these elements are mainly polluted by humans. The enrichment factors of Br, Pb, Sb and Zn are basically greater than 25, and the enrichment factors of these elements are 246.43 ± 168.81, 133.28 ± 115.03, 403.305 ± 396.xviii and ninety.67 ± 67.01, respectively. Especially for Br and Sb, the enrichment factors are all more than 100, and the enrichment is very serious, indicating that these elements are particularly polluted by humans; Zn, Br, and Lead mainly come from coal combustion, machine frazzle, garbage incineration and other man-made pollution [19,30]. It tin be seen that during the sampling menses, Zn, Lead and other elements are seriously polluted by coal combustion, machine exhaust and garbage incineration.
iii.5. Inferences most Emission Sources
The main piece of work of this report is to observe the environmental sample PM2.five based on FTIR, IC and XRF, and conduct multiple analyses combined with the atmospheric environmental data from Tianyun big information website [23] to determine the chemical limerick and master source of atmospheric particulate matter. The chemical limerick of atmospheric particulate matter is qualitatively analyzed by FTIR to determine the general types of collected samples. The specific components in the collected samples are quantitatively analyzed past IC and XRF, and the changes of pollution sources during sampling are further evaluated past enrichment gene analysis.
At the aforementioned time, the concentrations of CO, And soii and NOtwo from Tianyun big data website [23] are analyzed to obtain the general characteristics of particulate pollution. It can be seen from Effigy 8 that the peak concentration of CO, SO2 and NOii has decreased slightly since Apr. The main sources are industrial and mining enterprises, transportation, household stoves, heating boilers, setting off fireworks, charcoal burning and smoking. Based on the previous assay of PM2.v emission sources in Wuhan, combined with the relevant information of Wuhan Environmental Quality annual report, it tin can be inferred that most of CO, SO2 and NO2 in Wuhan'southward atmosphere may be generated by transportation. This is very consistent with the results of the enrichment gene analysis. Elements such equally Br, Pb, and Zn are the master enriched elements during the sampling menses. The enrichment factor has been at a high level (in a higher place 100). The high enrichment of these elements as well reflects the mechanism. The exhaust emissions of motor vehicles crusade serious pollution to the environment. At the same time, during this period, the concentration of PM2.5 was basically maintained at about fifty μg/mthree, and the concentration of PM10 was basically kept below 110 μg/m3, and the environmental quality was relatively good.
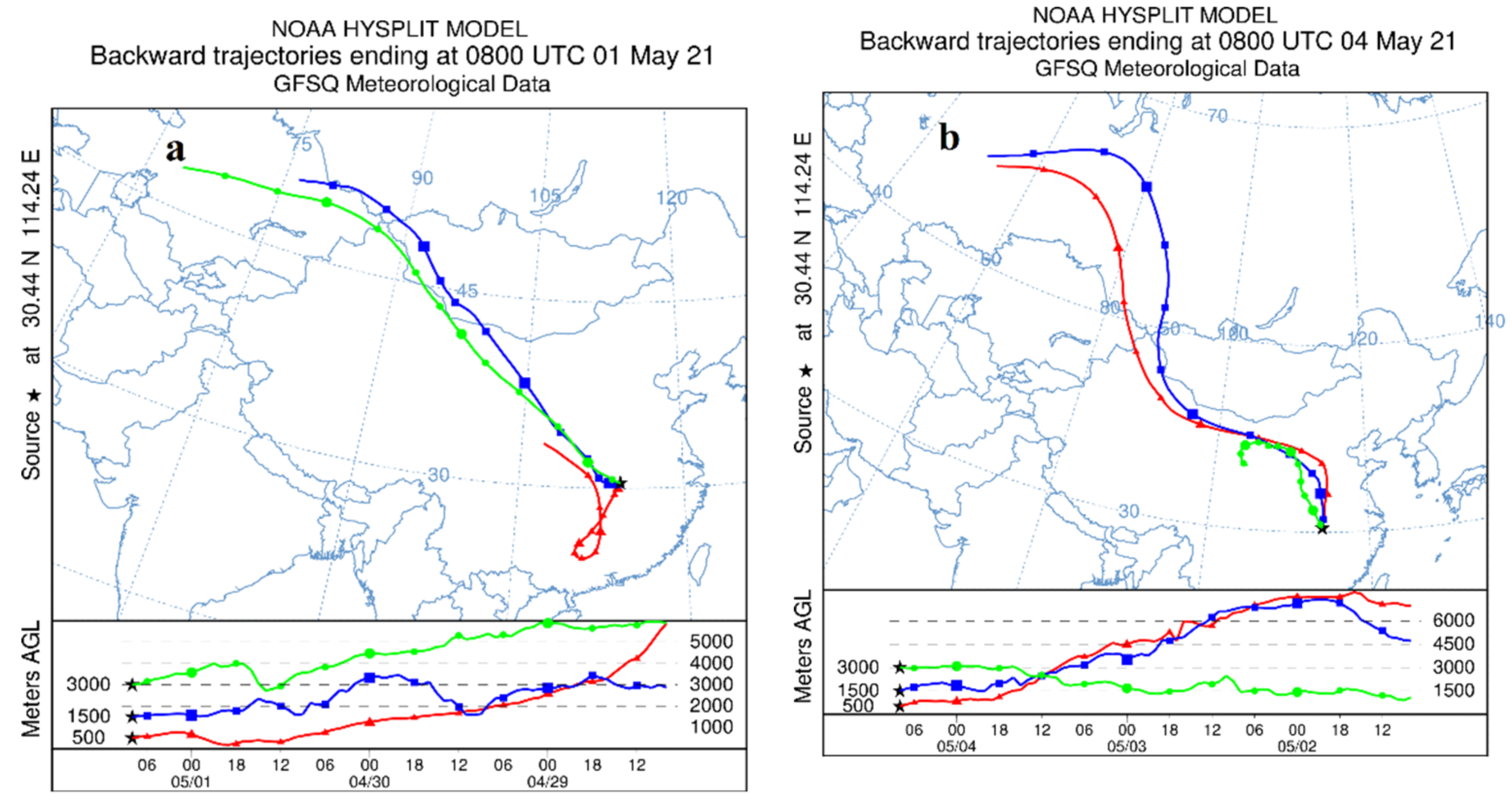
In gild to farther determine the source of aerosol particulate matter, the online model of HYSPLIT [31,32] was used to conduct a 72-h backward trajectory assay to survey the source of atmospheric particulate matter in Wuhan on i May 2021 (Effigy 9a) and 4 May 2021 (Figure 9b). The backward trajectory is shown in Figure nine. Through the backward trajectory diagram, it can exist institute that during the sampling period, Wuhan was mainly affected by the air currents from the Siberia, Inner Mongolia, and Loess Plateau regions of Russia. The bare land in the Siberia region of Russia and the sandstorm in the north may be affected. Information technology is the primary source of soil dust. At the same fourth dimension, the hot and humid air from the south volition also bring some pollutants to the Wuhan surface area. During this period, the enrichment of Mg, Sc, Ni, Cu and other elements may also be related to this.
4. Conclusions
The aim of this study is to investigate the chemic composition and characterization for PM2.five from Huangjiahu expanse of Wuhan city, some experimental setups, i.e., FTIR, IC, and XRF have been used. The sampling period is from 26 April to 24 May 2021. Likewise coupling with Wuhan'southward atmospheric environmental data and the chemical composition of particulate matter, the changes of emission sources have been analyzed. The followings are the main conclusions:
(one) During the sampling period, the concentration range of PM2.5 was 33.52 ± viii.5 μg/1000three, and the concentration range of PM10 was 81.0 ± 32 μg/m3. The results prove that the air quality in Wuhan was well during the sampling period.
(two) According to the measurement and analysis information of FTIR and XRF instruments, the collected samples are mainly equanimous of organic affair, inorganic ions, oxides, and xx kinds of elements (C, O, Na, Mg, Al, Si, P, South and other elements), which plainly come up from coal combustion, industrial pollutant emissions and automobile exhaust.
(three) Through the analysis of the oxidation efficiency of SO4 2−, the results show that the oxidation efficiency in the temper is low during the sampling period, indicating that the photochemical reaction is weak. Correlation analysis of SO4 2−/NO3 − shows that the sources of So4 2−, NOthree − are consistent, and the main source is the burning of fossil fuel. Meanwhile, the assay results of the ratio of the mass concentrations of the 2 show that during the sampling menses, the mobile source emission sources are less than the stationary source emission sources.
(4) The enrichment factor analysis method has been provided, and the aim is to clarify chief enriched elements, such as Br, Lead, Sb, Zn based on XRF measurement. The enrichment factors of these elements range are 246.43 ± 168.81, 133.28 ± 115.03, 403.305 ± 396.xviii, 90.67 ± 67.01 respectively. The results testify that these elements were extremely polluted by humans during the sampling menstruation, and their main source was vehicle exhaust emissions.
Author Contributions
Conceptualization, M.C.; investigation, F.C.; methodology, F.C.; writing—original draft, Q.Chiliad.; writing—review & editing, G.C. All authors accept read and agreed to the published version of the manuscript.
Funding
This piece of work was supported by the National Natural Science Foundation of China (Grant No. 51876147).
Institutional Review Board Statement
Not applicative.
Informed Consent Statement
Non applicable.
Data Availability Statement
Not applicable.
Acknowledgments
This work was supported by the National Natural Science Foundation of China (Grant No. 51876147). The authors gratefully acknowledge the NOAA Air Resources Laboratory (ARL) for the provision of the HYSPLIT transport and dispersion model, equally well every bit the Gear up website.
Conflicts of Interest
The authors declare no disharmonize of interest.
References
- He, Z.; Dong, C.; Liang, D.; Mao, J. A weightedsum-of-gray soot-fractal-aggregates model for nongray heat radiations in the loftier temperature gassoot mixture. J. Quant. Spectrosc. Radiat. Transf. 2021, 260, 107431. [Google Scholar] [CrossRef]
- Mao, Q.; Huang, C.; Chen, Q.; Zhang, H.; Yuan, Y. Satellite-based identification of aerosol particle species using a 2d-space aerosol nomenclature model. Atmos. Environ. 2019, 219, 117057. [Google Scholar] [CrossRef]
- Mao, Q. Recent developments in geometrical configurations of thermal energy storage for concentrating solar power constitute. Renew. Sustain. Energy Rev. 2016, 59, 320–327. [Google Scholar] [CrossRef]
- Mao, Q.; Zhang, H.; Chen, Q.; Huang, C.; Yuan, Y. Satellite-based cess of directly droplets radiative forcing using a Look-upward table established through AERONET observations. Infrared Phys. Technol. 2019, 102, 103017. [Google Scholar] [CrossRef]
- Mao, Q.; Huang, C.; Zhang, H.; Chen, Q.; Yuan, Y. Operation of MODIS Aerosol Products under annual/seasonal scales and different air conditions over Communist china. Sci. China Technol. Sci. 2021, 64, 774–784. [Google Scholar] [CrossRef]
- Bu, X.; Xie, Z.; Liu, J.; Wei, 50.; Wang, X.; Chen, M.; Ren, H. Global PM2.v-attributable wellness brunt from 1990 to 2017: Estimates from the Global Brunt of illness study 2017. Environ. Res. 2021, 197, 111–123. [Google Scholar] [CrossRef]
- Chai, G.; He, H.; Sha, Y.; Zhai, G.; Zong, S. Effect of PM2.five on daily outpatient visits for respiratory diseases in Lanzhou, China. Sci. Total Environ. 2019, 649, 1563–1572. [Google Scholar] [CrossRef] [PubMed]
- Jain, V.; Dey, S.; Chowdhury, S. Ambient PM2.5 exposure and premature mortality burden in the holy city Varanasi, Republic of india. Environ. Pollut. 2017, 226, 182–189. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.; Pang, Y.; Luo, X.; Chen, Q.; Wu, Fifty.; Tang, M.; Hong, Y.; Chen, J.; Jin, Fifty. The cytotoxicity and genotoxicity of PM2.5 during a snow issue in different functional areas of a megacity. Sci. Full Environ. 2020, 741, 140267. [Google Scholar] [CrossRef] [PubMed]
- Santoso, 1000.; Lestiani, D.D.; Damastuti, E.; Kurniawati, Due south.; Kusmartini, I.; Atmodjo, D.P.D.; Sari, D.K.; Muhtarom, T.; Permadi, D.A.; Hopke, P.Thousand. Long term characteristics of atmospheric particulate matter and compositions in Jakarta, Indonesia. Atmos. Pollut. Res. 2020, 11, 2215–2225. [Google Scholar] [CrossRef]
- Zhang, C.; Zou, Z.; Chang, Y.; Zhang, Y.; Wang, Ten.; Yang, X. Source cess of atmospheric fine particulate affair in a Chinese megacity: Insights from long-term, high-time resolution chemic composition measurements from Shanghai flagship monitoring supersite. Chemosphere 2020, 251, 126598. [Google Scholar] [CrossRef]
- Li, X.; Yan, X.; Wu, T.; Zhang, X.; Yu, H. Risks and phyto-uptake of micro-nano size particulates spring with potentially toxic metals in Pb-contaminated alkaline soil (NW China): The role of particle size fractions. Chemosphere 2021, 272, 129508. [Google Scholar] [CrossRef] [PubMed]
- Xie, C.; Yan, 50.; Liang, A.; Che, Due south. Understanding the washoff processes of PM2.five from leaf surfaces during rainfall events. Atmos. Environ. 2019, 214, 116844. [Google Scholar] [CrossRef]
- Khuzestani, R.B.; Schauer, J.J.; Wei, Y.; Zhang, Y.; Zhang, Y. A non-destructive optical color infinite sensing organization to quantify elemental and organic carbon in atmospheric particulate matter on Teflon and quartz filters. Atmos. Environ. 2017, 149, 84–94. [Google Scholar] [CrossRef]
- Chang, D.Y.; Lelieveld, J.; Steil, B.; Yoon, J.; Yum, S.S.; Kim, A.-H. Variability of aerosol-deject interactions induced by different cloud droplet nucleation schemes. Atmos. Res. 2021, 250, 105367. [Google Scholar] [CrossRef]
- Cheng, H.; Wang, Z.; Feng, J.; Chen, H.; Zhang, F.; Liu, J. Carbonaceous species composition and source circulation of PM (2.5) in urban atmosphere of Wuhan. Ecol. Environ. 2012, 21, 1574–1579. (In Chinese) [Google Scholar]
- Querol, 10.; Zhuang, X.; Alastuey, A.; Viana, M.; Lv, Due west.; Wang, Y.; Lopez, A.; Zhu, Z.; Wei, H.; Xu, S. Speciation and sources of atmospheric aerosols in a highly industrialised emerging mega metropolis in key Red china. J. Environ. Monit. 2016, 8, 1049–1059. [Google Scholar] [CrossRef]
- Huang, F.; Zhou, J.; Li, H.; Chen, N. Analysis of Causes and Sources of Typical Air Pollution Events in Summer and Winter in Wuhan Metropolis. Res. Environ. Sci. 2020, 33, 280–288. (In Chinese) [Google Scholar]
- Chen, Q.; Huang, C.; Xiao, T.; Yuan, Y.; Mao, Q.; Tan, H. Characterization of atmospheric aerosols and source apportionment analyses in urban Harbin, northeast Mainland china. Infrared Phys. Technol. 2019, 103, 103109. [Google Scholar] [CrossRef]
- Zhang, Y.; Lang, J.; Cheng, S.; Li, S.; Zhou, Y.; Chen, D.; Zhang, H.; Wang, H. Chemical composition and sources of PM1 and PM2.5 in Beijing in autumn. Sci. Total Environ. 2018, 630, 72–82. [Google Scholar] [CrossRef]
- Elcoroaristizabal, S.; Amigo, J.M. Near infrared hyperspectral imaging every bit a tool for quantifying atmospheric carbonaceous aerosol. Microchem. J. 2021, 160, 105619. [Google Scholar] [CrossRef]
- Coury, C.; Dillner, A.M. ATR-FTIR characterization of organic functional groups and inorganic ions in ambient aerosols at a rural site. Atmos. Environ. 2009, 43, 940–948. [Google Scholar] [CrossRef]
- Tianyun Big Information Website. Available online: https://world wide web.ebd120.com/ (accessed on 30 May 2021).
- Li, Ten.; Zhang, T.; Sun, F.; Song, X.; Zhang, Y.; Huang, F.; Yuan, C.; Yu, H.; Zhang, G.; Qi, F.; et al. The relationship between particulate matter retentiveness chapters and leafage surface micromorphology of ten tree species in Hangzhou, China. Sci. Total Environ. 2021, 771, 144812. [Google Scholar] [CrossRef]
- Kumar, S.; Raman, R.Southward. Inorganic ions in ambient fine particles over a National Park in primal India: Seasonality, dependencies between SO4 ii−, NO3 −, and NH4 +, and neutralization of aerosol acerbity. Atmos. Environ. 2016, 143, 152–163. [Google Scholar] [CrossRef]
- Zhao, K.; Liu, Ten.; Hui, L.; He, Q.; Zhong, Y.; Yan, J. Characteristics and Sources of Water-Soluble Ions of PM2.5 in Winter in Urumqi. J. Desert Res. 2015, 35, 707–714. (In Chinese) [Google Scholar]
- Niu, Y.; Wang, F.; Liu, S.; Zhang, Due west. Source analysis of heavy metal elements of PM2.5 in bottle in a academy in winter. Atmos. Environ. 2021, 244, 117879. [Google Scholar] [CrossRef]
- Cable, E.; Deng, Y. Trace Elements in Atmospheric Wet Atmospheric precipitation in the Detroit Metropolitan Area: Levels and Possible Sources. Chemosphere 2018, 210, 1091–1098. [Google Scholar] [CrossRef] [PubMed]
- Cáceres, J.O.; Sanz-Mangas, D.; Manzoor, S.; Pérez-Arribas, Fifty.V.; Anzano, J. Quantification of particulate affair, tracking the origin and relationship between elements for the ecology monitoring of the Antarctic region. Sci. Total Environ. 2019, 665, 125–132. [Google Scholar] [CrossRef]
- Liu, L.; Liu, Y.; Wen, W.; Liang, L.; Ma, X.; Jiao, J.; Guo, Yard. Source identification of trace elements in PM2.5 at a rural site in the north cathay plain. Atmosphere 2020, eleven, 179. [Google Scholar] [CrossRef]
- Rolph, G.; Stein, A.; Stunder, B. Real-time Environmental Applications and Display organisation: Ready. Environ. Model. Softw. 2017, 95, 210–228. [Google Scholar] [CrossRef]
- Real-Fourth dimension Environmental Application and Display Organization. Available online: https://www.prepare.noaa.gov/index.php (accessed on 4 May 2021).
Figure 1. The specific orientation of the sampling point.
Figure 1. The specific orientation of the sampling bespeak.
Effigy 2. The boilerplate monthly concentrations of PM2.5 and PM10 in Wuhan from Jan 2017 to May 2021 (a), and the average daily concentrations of PM2.five and PM10 in Wuhan from January to May 2021 (b), nerveless from Tianyun big data website [23].
Figure two. The average monthly concentrations of PM2.five and PM10 in Wuhan from January 2017 to May 2021 (a), and the average daily concentrations of PM2.5 and PM10 in Wuhan from January to May 2021 (b), collected from Tianyun large data website [23].
Effigy iii. Infrared spectrograph results of particles collected on 28 Apr, xxx Apr and half-dozen May in 2021 in Wuhan.
Effigy iii. Infrared spectrograph results of particles collected on 28 April, 30 April and 6 May in 2021 in Wuhan.

Figure 4. Concentration data of four ions during sampling.
Effigy iv. Concentration data of four ions during sampling.

Figure v. The ratio of the mass concentration of Theniv 2− to NO3 − during sampling.
Effigy 5. The ratio of the mass concentration of Then4 2− to NOiii − during sampling.

Figure 6. Proportion of Si, Ca, Iron, S, K and other trace elements (a) and specific proportion of other trace elements (b).
Effigy six. Proportion of Si, Ca, Fe, S, Chiliad and other trace elements (a) and specific proportion of other trace elements (b).
Effigy 7. Results of enrichment factor analysis based on the XRF data.
Figure 7. Results of enrichment factor analysis based on the XRF data.
Figure 8. Variation diagram of CO, SO2 and NOii mass concentration during sampling.
Figure eight. Variation diagram of CO, And then2 and NOii mass concentration during sampling.
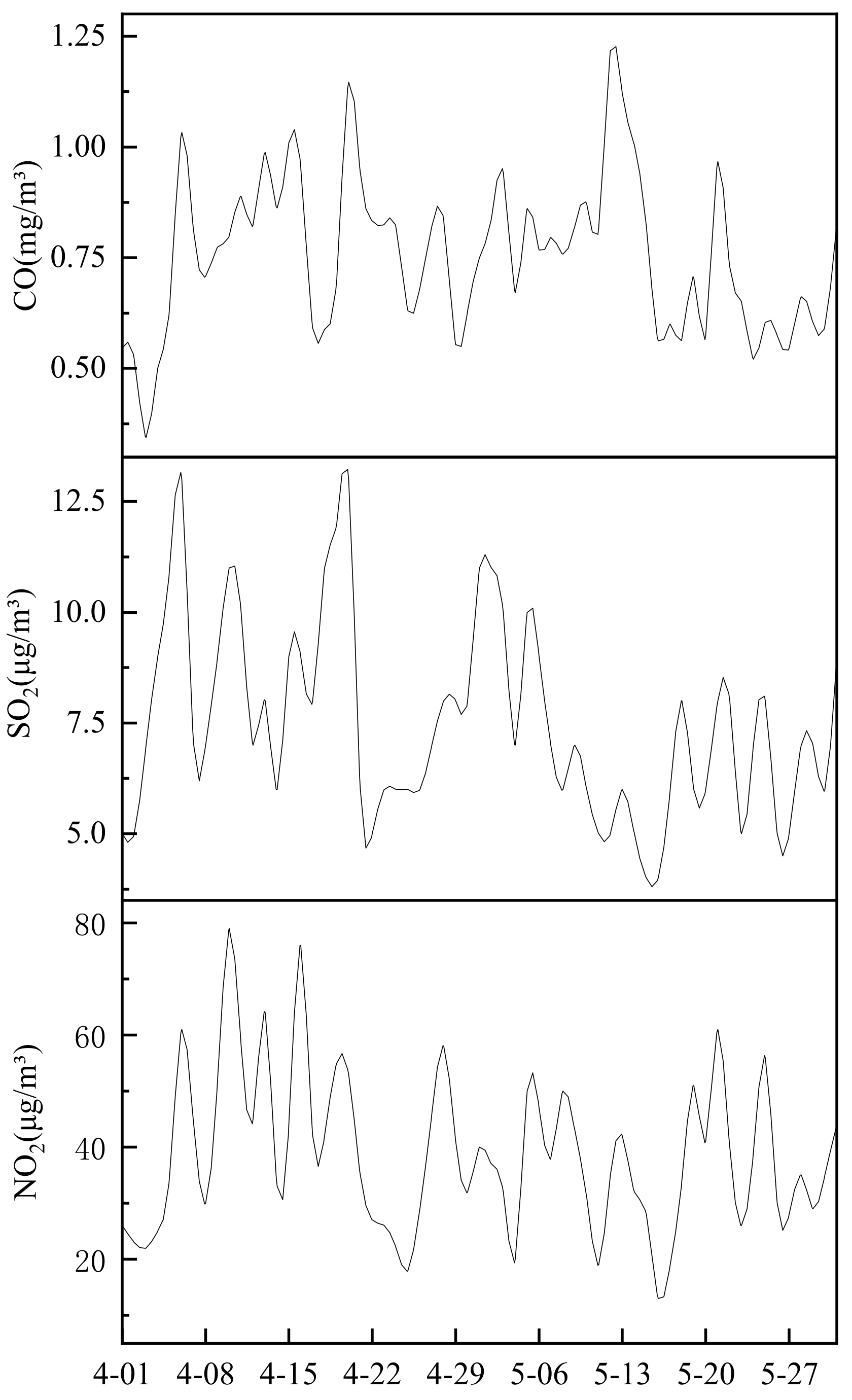
Figure 9. The 72-h astern trajectory tracking analysis results for Wuhan using the HYSPLIT model (web-version) on (a) 1 May, and (b) iv May 2021. The reddish, blue, and light-green lines represent the trajectories of different high airflow sources (500, 1500, 3000) in the report area.
Figure 9. The 72-h backward trajectory tracking analysis results for Wuhan using the HYSPLIT model (spider web-version) on (a) 1 May, and (b) iv May 2021. The red, blue, and greenish lines correspond the trajectories of different high airflow sources (500, 1500, 3000) in the study area.
Tabular array i. Cheque list of sample numbers (No.) and sampling date (SD).
Table 1. Check list of sample numbers (No.) and sampling engagement (SD).
| No. | SD | No. | SD | No. | SD | No. | SD |
|---|---|---|---|---|---|---|---|
| 1 | 26 April 2021 | 4 | 29 April 2021 | 7 | six May 2021 | 10 | 9 May 2021 |
| 2 | 27 April 2021 | 5 | 30 April 2021 | eight | 7 May 2021 | 11 | 20 May 2021 |
| 3 | 28 April 2021 | 6 | i May 2021 | 9 | 8 May 2021 | 12 | 24 May 2021 |
Table 2. Standard curve of four anions.
Table 2. Standard curve of 4 anions.
| F− | Cl− | So4 2− | NO3 − | |
|---|---|---|---|---|
| Standard curve line | y = 0.4549x + 0.0004 | y = 0.2753x | y = 0.1945x | y = 0.6606x |
| Correlation coefficient | r = 0.9998 | r = 0.9999 | r = 0.9998 | r = 0.9998 |
| Publisher's Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and atmospheric condition of the Artistic Commons Attribution (CC By) license (https://creativecommons.org/licenses/by/iv.0/).
Source: https://www.mdpi.com/2073-4433/12/11/1393/htm
0 Response to "Real-time Chemical Characterization of Atmospheric Particulate Matter in China a Review"
Post a Comment